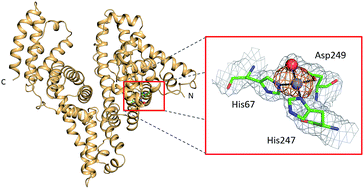
Circulatory zinc transport is controlled by distinct interdomain sites on mammalian albumins… The major site in both HSA and ESA has tetrahedral geometry and comprises three protein ligands from the sidechains of His67, His247 and Asp249 and a water molecule. Isothermal titration calorimetric studies of a HSA H67A mutant confirm this to be the highest affinity Zn2+ site. Furthermore, analysis of Zn2+ binding to HSA and ESA proved the presence of secondary sites with 20–50-fold weaker affinities, which may become of importance under particular physiological conditions. Both calorimetry and crystallography suggest that ESA possesses an additional site compared to HSA, involving Glu153, His157 and His288. The His157 residue is replaced by Phe in HSA, incapable of metal coordination. Collectively, these findings are critical to our understanding of the role serum albumin plays in circulatory Zn2+ handling and cellular delivery.
Researchers capture first images of a process vital for wellbeing. Zinc is essential for wound healing, vision, DNA creation, for our senses of taste and smell, even for sexual health. Scientists have never fully understood the mechanism that moves the mineral through the body – until now. An international team has, for the first time, created detailed blueprints of the molecular moving vans that ferry this important mineral everywhere it’s needed through the blood.
Zinc is carried through the body by a protein known as serum albumin. Scientists had expected there would be a primary binding site where serum albumin binds with zinc. But the team also found several more secondary binding sites, revealing a more complex interaction than anticipated. “It’s different than it was predicted before,” said Katarzyna B. Handing, PhD.
While computer models previously had been used to predict how serum albumin picks up zinc, Minor’s team used Xray crystallography to create colorful images of zinc actually bound to serum albumin. The technique allows them to pinpoint the location of each particular zinc atom. It was a challenging task, but the resulting schematics allow scientists to see, for the first time, exactly how serum albumin and zinc come together.
With the finding, scientists have a better grasp of how the body maintains homeostasis. It’s a complex dance made all the more complicated by the fact that serum albumin also transports many other things, such as hormones and fatty acids. “Homeostasis is extremely important, and it can be affected by the level of zinc you are taking into your body. But it can be also affected by other elements,” Handing said. “If you have an elevated level of fatty acids, eg as a result of diabetes or obesity, the zinc homeostasis can be disturbed.”

An artist’s interpretation of zinc being transported in the bloodstream. Credit: Courtesy Wladek Minor
This is important because the body needs zinc, but too much zinc is toxic. So the body must make it available where it is needed, but, at the same time, it must prevent excessive buildup. If something goes wrong with the zinc regulation process, that can have a ripple effect, throwing the body’s delicate balances out of whack and potentially having serious effects on health.
Ivan G. Shabalin, PhD noted the research could help shed light on why certain drugs affect some patients differently than others. “We are going towards an understanding of all these complex relationships,” he said. “You have this one molecule [serum albumin], and you have hundreds – possibly thousands – of different molecules which bind to it. We need to understand all this interplay. By studying zinc binding to albumin, we are understanding this relationship deeper.” http://pubs.rsc.org/en/Content/ArticleLanding/2016/SC/C6SC02267G!divAbstract https://www.eurekalert.org/pub_releases/2016-11/uovh-hhy110316.php






Recent Comments