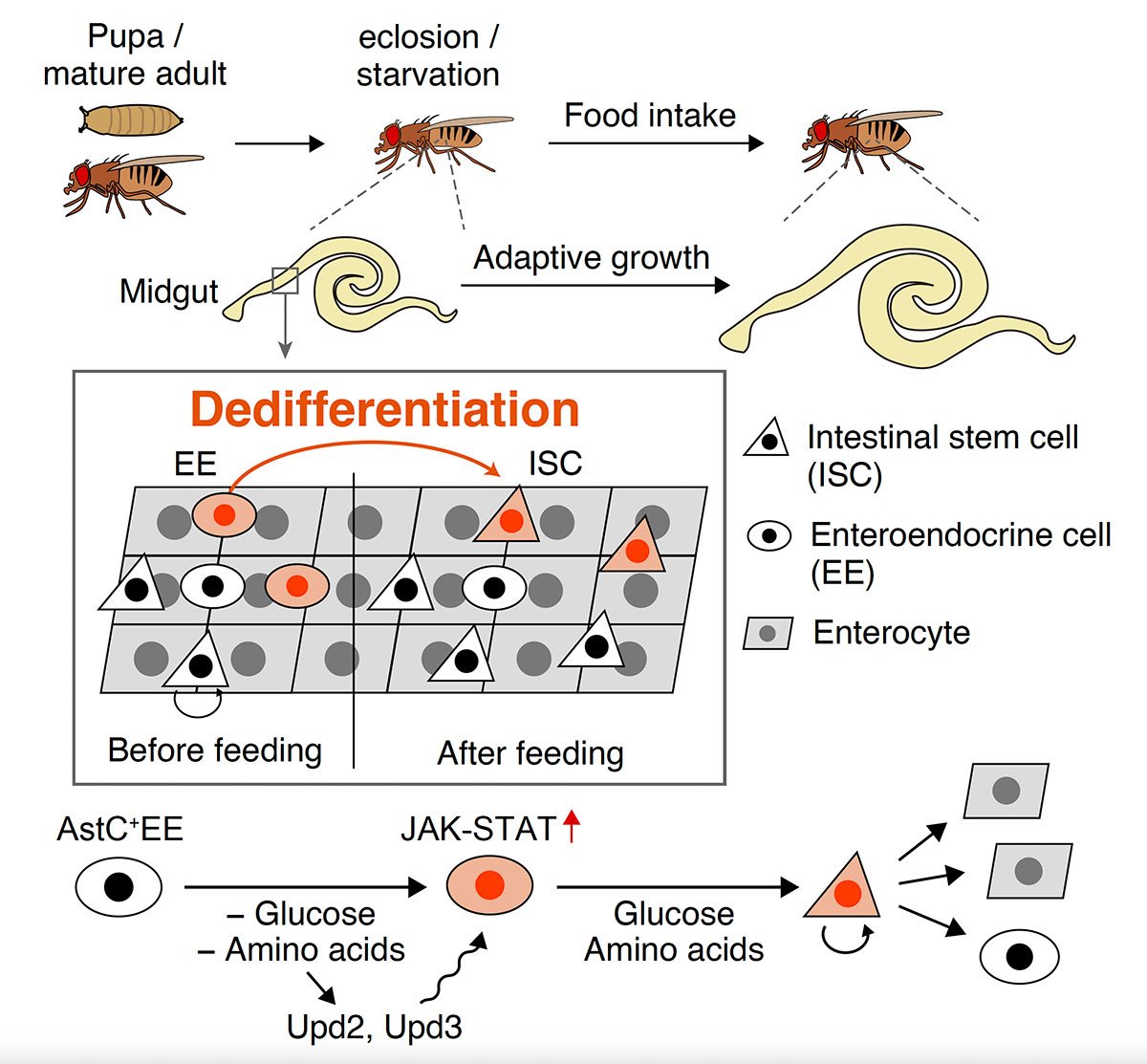
Researchers have unveiled an intriguing phenomenon of cellular reprogramming in mature adult organs, shedding light on a novel mechanism of adaptive growth. The study, which was conducted on fruit flies (Drosophila), provides further insights into dedifferentiation — where specialized cells that have specific functions transform into less specialized, undifferentiated cells like stem cells.
Until now, dedifferentiation has primarily been associated with severe injuries or stressful conditions, observed during tissue regeneration and diseases like tumorigenesis. However, the researchers have unearthed a previously unknown facet: enteroendocrine cells (EEs) within the intestinal epithelium undergo dedifferentiation into intestinal stem cells (ISCs) in response to nutritional changes, such as recovery from starvation.
“Through meticulous experimentation, we identified a subset of enteroendocrine cells residing in the adult midgut of Drosophila, which exhibit dedifferentiation into ISCs when nutrient levels fluctuate,” states Hiroki Nagai, first author of the study and a postdoc who was previously based at Tohoku University’s Frontier Research Institute for Interdisciplinary Sciences (FRIS). “By utilizing in vivo lineage tracing of EEs and single-cell RNA sequencing, we pinpointed the dedifferentiating EE subpopulation and developed a genetic system for selectively removing ISCs derived from dedifferentiation, a process known as ablation.”
Remarkably, the ablation experiments demonstrated that dedifferentiation is vital for ISC expansion and subsequent intestinal growth following food intake. Previous studies using mice relied on massive stem cell ablation to induce dedifferentiation. Yet, in the current research, stem cells were not lost but instead increased in response to nutritional stimuli. This crucial distinction demonstrates that dedifferentiation is not limited to regenerative contexts but significantly contributes to organ remodeling during environmental adaptations.
Furthermore, the team unraveled the molecular mechanism driving nutrient-dependent dedifferentiation: a deficiency in dietary glucose and amino acids activates the JAK-STAT signaling pathway in EEs, facilitating the conversion of EEs into ISCs during post-starvation recovery. When combined with findings from other studies, this implies that the nutrient-dependent dedifferentiation could be an evolutionary conserved mechanism across species.
Yuichiro Nakajima, also formerly based at FRIS and corresponding author of the paper, states that this could lead to being able to control artificial cellular reprogramming in vivo. “If we figure out specific nutrients and the detailed signaling that induce dedifferentiation, we could control cell fate plasticity by nutritional intervention and/or pharmacological treatments”
Looking ahead, they hope to focus on examining cell fate plasticity under physiological conditions beyond nutrition, such as reproduction, temperature, light, and exercise. Doing so may uncover novel mechanisms underlying environmental adaptations. http://www.tohoku.ac.jp/en/press/nutrients_drive_cellular_reprogramming_in_the_intestine.html






Recent Comments