
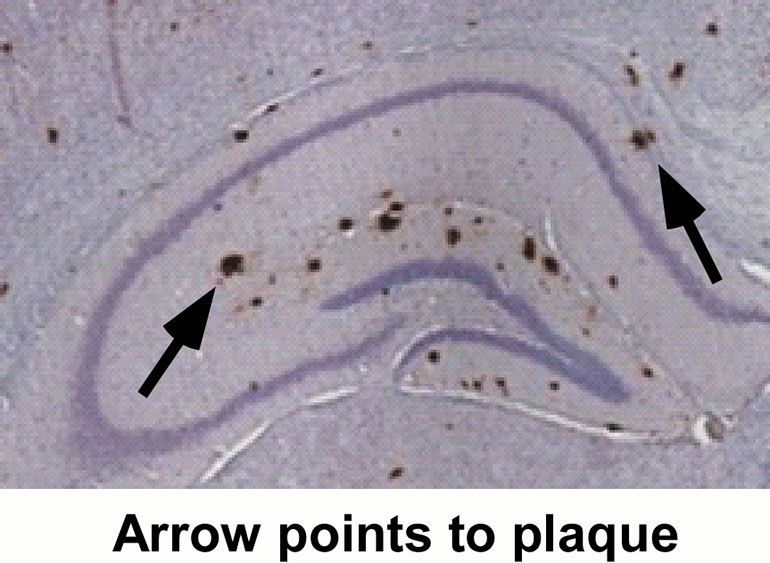
Images that predate the formation of toxic clumps of beta-amyloid, the protein believed to be at the root of Alzheimer’s disease, have now been captured by researchers. Credit: Illustration: Per Uvdal
MAX IV synchrotron in Lund, Sweden – the strongest of its kind in the world – has been used to produce images that predate the formation of toxic clumps of beta-amyloid, the protein believed to be at the root of Alzheimer’s disease. The unique images appear to contradict a previously unchallenged consensus. Instead of attempting to eliminate beta-amyloid plaques, the researchers now suggest stabilizing the protein.
It is a long-held belief in the scientific community that the beta-amyloid plaques appear almost instantaneously. Hence the term “popcorn plaques...
Read More






Recent Comments