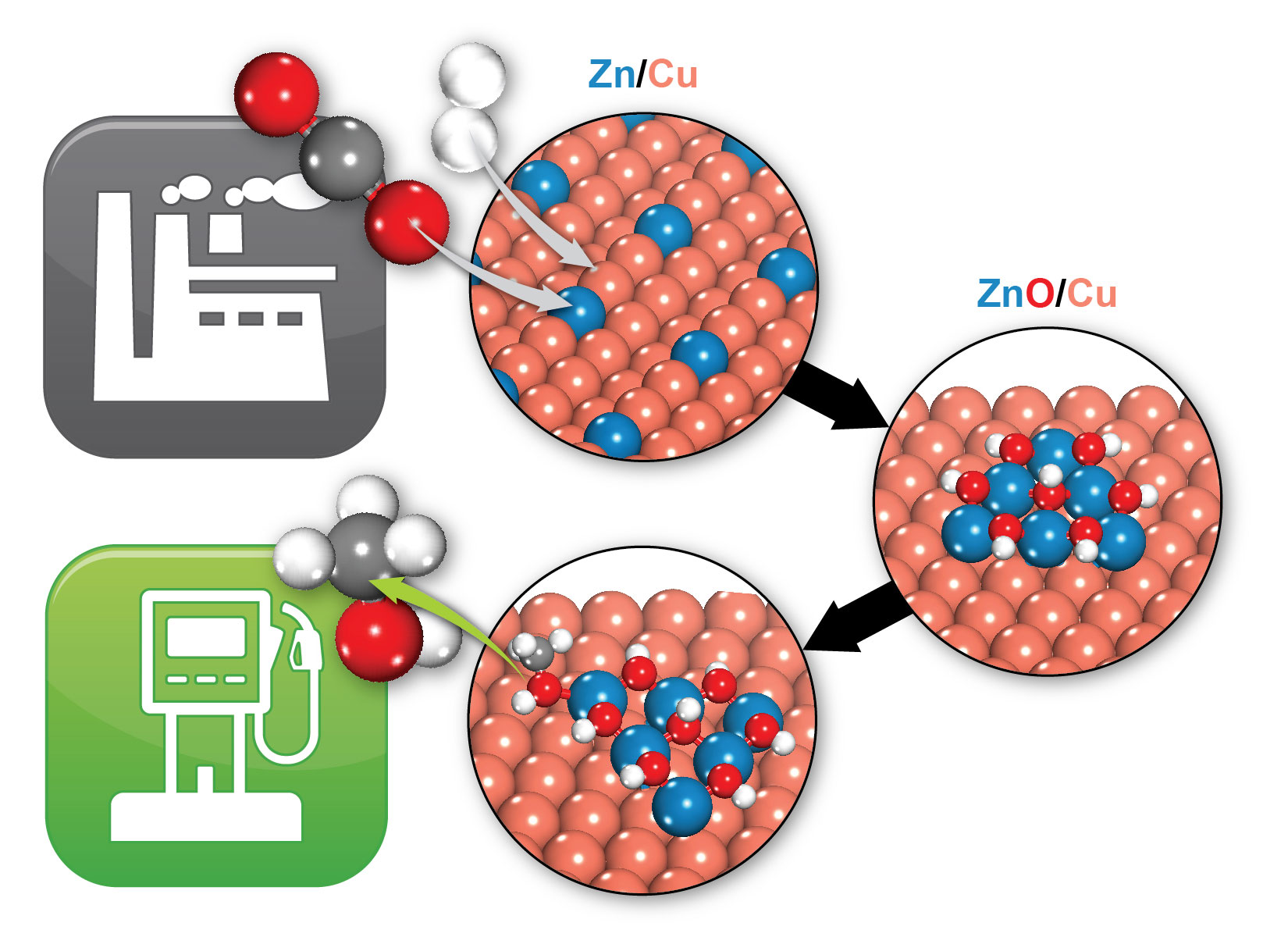
Brookhaven scientists identified how a zinc/copper (Zn/Cu) catalyst transforms carbon dioxide (two red and one grey balls) and hydrogen (two white balls) to methanol (one grey, one red, and four white balls), a potential fuel. Under reaction conditions, Zn/Cu transforms to ZnO/Cu, where the interface between the ZnO and Cu provides the active sites that allow the formation of methanol.
Results will guide design of improved catalysts for transforming pollutant to useful chemicals. Capturing CO2 and converting it to useful chemicals such as methanol could reduce both pollution and our dependence on petroleum products. So scientists are intensely interested in the catalysts that facilitate such chemical conversions...
Read More



Recent Comments